 |
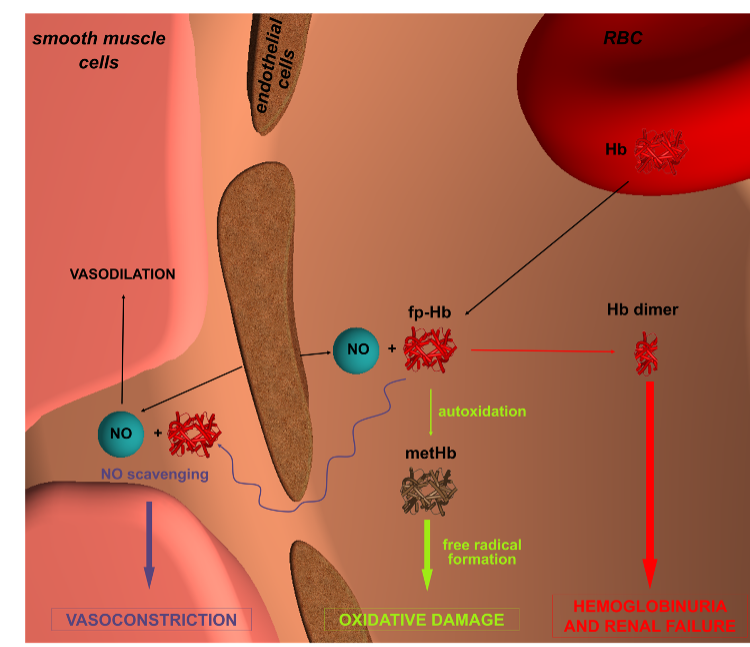
Hemolysis is the abnormal breakdown of red blood cells (RBCs) resulting in the release of hemoglobin contained in them as free plasma hemoglobin (fp-Hb). This phenomenon entails an important clinical risk triggered by various causal agents and the growing number of acquired and iatrogenic hemolytic disorders invites us to consider hemolysis as a new mechanism of disease onset.
Hemoglobin within red blood cells is a tetramer, but during hemolysis it tends to form dimers. An equilibrium between Hb tetramers and dimers is therefore present in the plasma. Hb dimers, being smaller, can pass through the renal glomeruli and, when the absorption capacity is exceeded, hemoglobinuria appears, i.e. the presence of hemoglobin in the urine, which in the long run causes renal failure. Furthermore, free hemoglobin can migrate into the endothelium of blood vessels and react with nitric oxide (NO), a vasodilator produced by the endothelium, causing vasoconstriction and hypertension.
|
|
Outside the red blood cells, the iron contained in hemoglobin within a cofactor called heme is oxidized (oxyhemoglobin, oxyHb becomes methemoglobin, metHb), can then be released into the plasma and promote phenomena such as lipid peroxidation, damage to DNA and proteins, thus causing oxidative damage.
Physiological systems have evolved to control plasma free hemoglobin levels when intravascular hemolysis occurs. For example, Hb dimers bind with high affinity to serum haptoglobin (Hp) and the complex is captured by the CD163 Hb receptor present on macrophages, internalized and degraded. However, the effectiveness of these strategies is often exceeded in pathological hemolytic conditions: since HP is not recycled, its rapid depletion is observed in severe hemolytic conditions.
|
 |
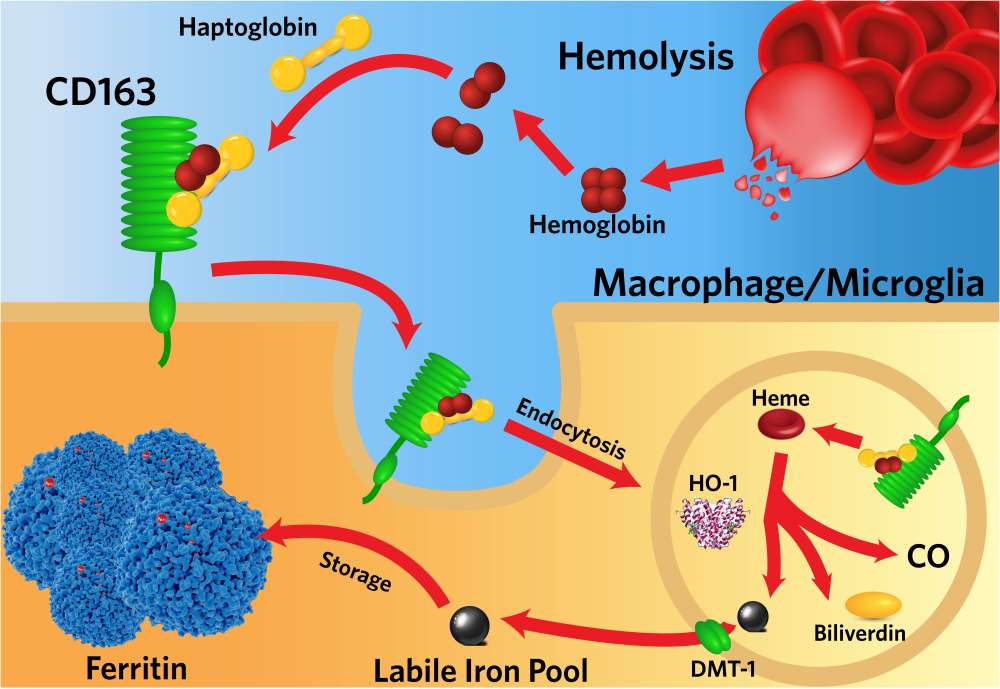 |
In case of hemolytic shock today the only therapeutic option is plasma exchange, while in chronic conditions hemolysis is monitored only as a risk factor for an unfavorable prognosis. The only therapeutic approach proposed to limit the effects of hemolysis is the administration of Hb, with the complications linked to the administration of proteins in massive doses, and in any case this approach implies that the organism is responsible for the disposal of high quantities of Hb, heme and iron.
|